The U.K. is rolling out a program in which all seriously ill children without a diagnosis will be able to get a full genetic work-up for themselves and their parents, for free. The blood test, called whole-genome sequencing, yields a Human Genome Project’s worth of information about an individual person.
It’s especially useful for children born with mysterious — but likely genetic — abnormalities that severely affect their development and quality of life.
Whole-genome sequencing is not available in Canada yet under any provincial health plan. But Ontario’s health-care quality agency is currently reviewing a proposal to cover it for children with unexplained developmental delay, said Wendy Ungar, director of technology assessment at Toronto’s SickKids hospital, a major Canadian centre for this area of research.
Dr. Stephen Scherer, director of the hospital’s centre for applied genomics, gets excited talking about a future, perhaps in as little as five years, in which whole-genome sequencing for kids and parents is a routine test in children’s hospitals. The information it yields can give clues about how a patient will respond to certain drugs and help predict how their disease will unfold, he said. Even kids who already have a diagnosis can get more information about the different genes involved, or what complicating factors might be at play. Plus, the same patient’s genome — data that only needs to be collected once — can be re-analyzed as new information becomes available.
“The U.K. has got the whole thing figured out. The latest news is quite incredible,” Scherer said. “We need a national strategy in this country, and we don’t have one.”
“The game-changer here is that with one test, we get all the genes,” he continued. “For better or worse, we get all of it.”
We need a national strategy in this country, and we don’t have one
For the non-scientist, it’s hard to imagine how there could be a “worse” side to more comprehensive, accurate medical testing. But the question of whether knowing all of a patient’s genetic information might be a bit of medical TMI is actually very controversial in the health-care world.
And the test does cost money. It’s not the year 2000 anymore, so it doesn’t take a decade and $3 billion to sequence all your genes — more like a few days and about $1,000 U.S. in a research setting.
Currently, patients are offered a series of conventional genetic tests that get more useful, but also pricier for the health-care system, as you go, said Robin Hayeems, an associate professor of health policy at the University of Toronto and a scientist who studies cost-effective health care at SickKids.
The cheapest test turns up a definitive diagnosis about 15 per cent of the time, and the most expensive, about 20 to 30 per cent. Whole-genome sequencing, which provides all the information the other tests do, plus more, cracks 30 to 40 per cent of cases. Scherer puts the number at 43 per cent. Based on sticker price, whole-genome sequencing would seem to be the better deal.
But the cost-benefit calculation gets complicated, because while sequencing can cut down on costs of testing, it may increase costs after diagnosis. Once a child is identified as having genetic markers that indicate, for instance, autism, they’ll be sent to more specialists and referred for more treatments — though hopefully ones that are personalized.
Then there’s the whole issue of incidental findings, which geneticists call “secondary variance.”
Everyone has some weird stuff — typos, essentially — in their DNA, and lot of the variation has unknown significance. But in two to five per cent of people, a whole-genome test reveals important, actionable medical information, such as the trait that guarantees you’ll develop Huntington’s disease, or a mutation in one of the BRCA genes associated with breast cancer, Scherer said.
Not all the Canadian studies using whole-genome sequencing also tell the participants if there are significant secondary findings, Scherer said, but the ones that do communicate them to patients via a genetic counsellor.
“Sometimes we find more than what we’re looking for,” Hayeems said. For example, she says: Say screening for developmental delay turns up a BRCA mutation in a child or their mother. That’s associated with a 50 to 80 per cent chance of developing breast cancer in people with a history of the disease. But we don’t know how dangerous it is for someone with no breast cancer in the family tree. Hayeems worries overuse of sequencing could lead over-testing, over-monitoring and ballooning costs.
Ontario will likely be the first province to offer whole-genome sequencing, and Hayeems stressed that its health ministry needs to take a “cautious, evidence-informed approach” about who to offer it to and when.
“Our technologies are extremely precious resources, and we have an extremely constrained health-care budget,” she said. “I don’t think racing is getting ahead … Letting the U.K. do it first — I think that’s OK! We can learn from their experience.”
Scherer was more gung-ho. Analysis of whole-genome sequences is getting better all the time, he said, and the most important thing that’s going to make it better is a bigger data set of genomes from healthy and sick people — like the one the U.K. is amassing by radically increasing access to testing.
I don’t think racing is getting ahead ... Letting the U.K. do it first — I think that’s OK! We can learn from their experience
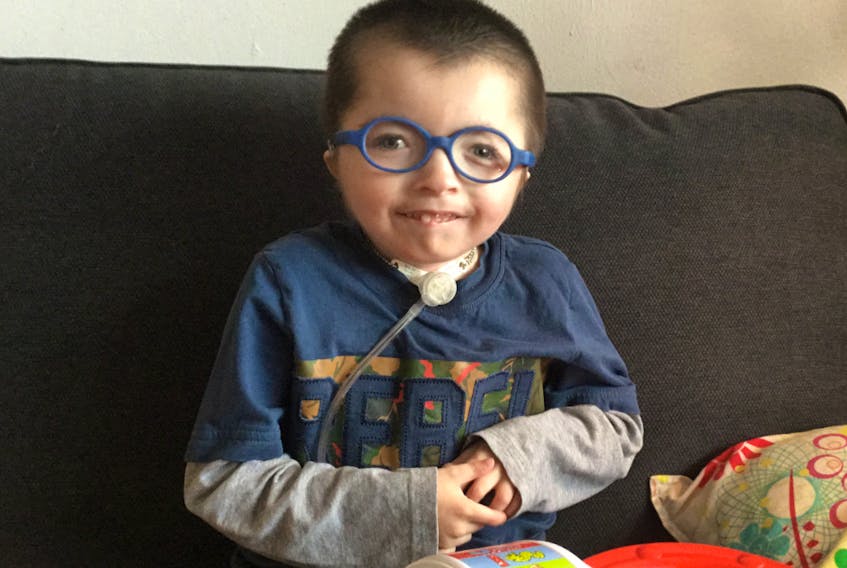
For the first eight years of her son Thomas’s life, Stephanie Stenabaugh constantly worried that his developmental delay and multiple medical issues were her fault.
She cried for days after learning, through a whole-genome sequencing study at SickKids, that he had a form of a rare genetic disease, Cornelia de Lange syndrome (CdLS).
“There was a great sense of relief … I, like every mother, tended to think, ‘What did I do? Maybe it was because I was working at the chemistry lab? Or because I was stressed? Was it my age?’ To realize that it was something that happened at conception, and it’s nothing that we as parents did … It would have been extremely beneficial to have that diagnosis (earlier),” she said.
Thomas, who died of a respiratory infection in April, a few months shy of his 10th birthday, weighed just 30 pounds — CdLS can cause a form of dwarfism — and needed a tracheaostomy tube to help him breathe. His speech was limited to a few hand signs for “more” and “mama,” but he had his ways of communicating.
“He had a speaking valve on his tube, so we could hear his laugh,” Stenabaugh
said. “He loved music. Anytime you played music, he would start swaying to the beat. I took him to the beach last summer, just me and him, and he loved it. He was such a happy kid.”
Although Thomas’s treatment plan didn’t change significantly after whole-genome sequencing, it helped Stenabaugh feel “less alone” and connect with other CdLS parents. She learned through them that it might be possible for Thomas to have a normal lifespan or even learn to walk.
He had shown signs of being able to stand, and Stenabaugh
wishes in hindsight that she pushed him a little harder. But she didn’t want to stress him out, and a doctor had expressed doubt he could even learn to hold his head up.
“I absolutely think (whole genome sequencing) should be covered,’ she said.
Scherer said he’s personally looking forward to living in a world with more answers for mysterious diseases. Because he’s always the one with his name in the news, Scherer often hears from people who are desperate to know what’s happening in their own or their child’s DNA.
“We’ll be using this as a molecular stethoscope.”
• Email: [email protected] | Twitter: genna_buck
Copyright Postmedia Network Inc., 2019